  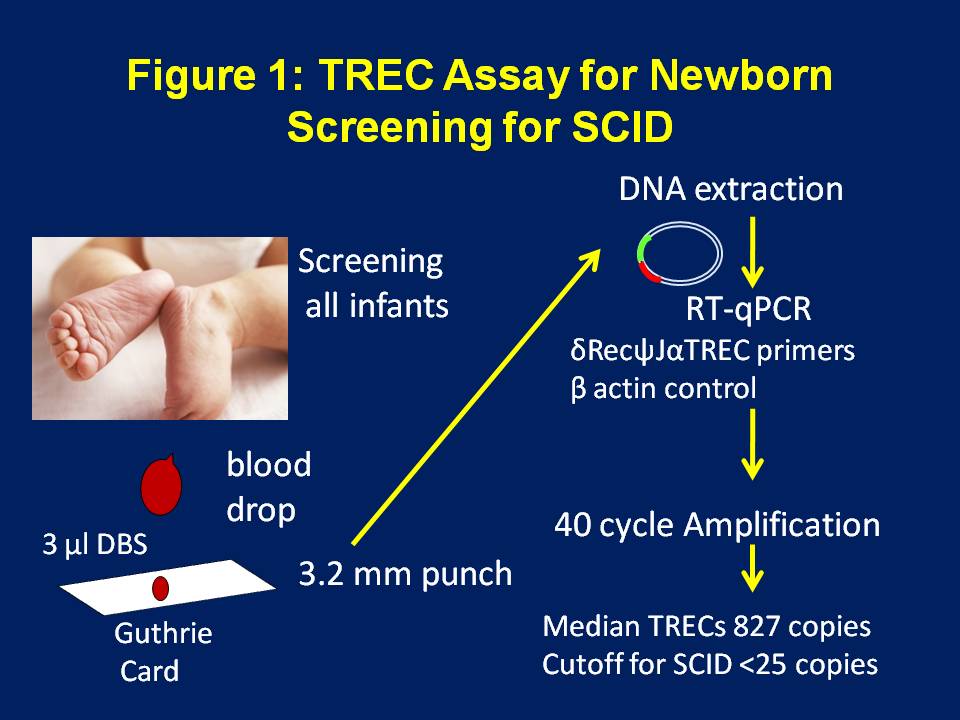
De novo variants may occur in ~50% of cases of X-linked SCID due to IL2RG pathogenic variants (Allenspach et al. Of note, a founder variant in DCLRE1C has been identified in the Navajo Native American population (Li et al. Pathogenic variants may be inherited or de novo. The frequency of the specific variants is dependent on the underlying genetic cause of disease. Large copy number events have also been reported in the causative genes for T cell negative SCID. Reported pathogenic variants in the causative genes include missense, nonsense, frameshift, splice site, and hypomorphic variants. Anywhere from ~13-31% of cases of SCID are idiopathic (Chan et al. Pathogenic variants in AK2, CD247, CD3D, CD3E, CORO1A, DCLRE1C (ARTEMIS), IL2RG, JAK3, LAT, LIG4, NHEJ1, PRKDC, PTPRC, RAG1, and RAG2 together are responsible for ~10% of SCID (Allenspach et al. Variants in IL7R make up ~5-10% of all SCID cases (Allenspach et al. Adenosine deaminase deficiency due to variants in ADA contributes to ~15-20% of SCID cases (Allenspach et al. All other genes in this panel show autosomal recessive inheritance. X-linked SCID caused by IL2RG pathogenic variants accounts for ~40-60% of all cases of SCID (Allenspach et al. The pattern of inheritance for SCID may be X-linked or autosomal recessive. To date, there are 17 genes in which pathogenic variants have been described as causative for T cell negative SCID (Bousfiha et al. This panel focuses on genetic causes of T cell negative SCID that have been identified through literature, OMIM, and HGMD searches. The four main categories of SCID, based upon which immune cells are defective (T, B, and/or NK cells), each have distinct genetic causes. Early hematopoietic stem cell transplantation (HSCT) is the most established treatment for patients with SCID (Kwan et al. In general, the prognosis is poor if there is a delay is diagnosis and therapy. The recurrence risk and prognosis may vary depending on the underlying genetics cause of the deficiency. Prior to NBS, population-based screening was the only method to identify those at risk for SCID prior to the onset of symptoms, however >80% of cases lack a positive family history (Chan and Puck. Diagnoses are typically made via utilization of flow cytometry-based testing and newborn screening (NBS) assessment of T cell receptor excision circles (TRECs), however it is necessary to establish a genetic diagnosis for genetic counseling, prognostication, and optimization of treatment (Kwan et al. The clinical features associated with SCID include recurrent and severe bacterial, viral, and fungal infections that begin in infancy (Fischer. The broad classification of SCID may be subdivided based on B cell status and further subdivided based on NK cell status, which may provide insight into the causative genetic defect (Kumrah et al. SCID is caused by genetic defects that inhibit lymphocyte development and function, resulting in no T cell differentiation and abnormal development of B and natural killer (NK) lymphocytes (Fischer. While the true incidence of SCID is unknown, newborn screening studies suggest that 1 in 70,000 births are affected with a range of 1 in 40,000 to 100,000 (Fischer. Severe combined immunodeficiency (SCID) encompasses a diverse group of rare, life-threatening disorders. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
